




High quality Hyaluronic Acid
suitable for pharmaceutical and medical
industries
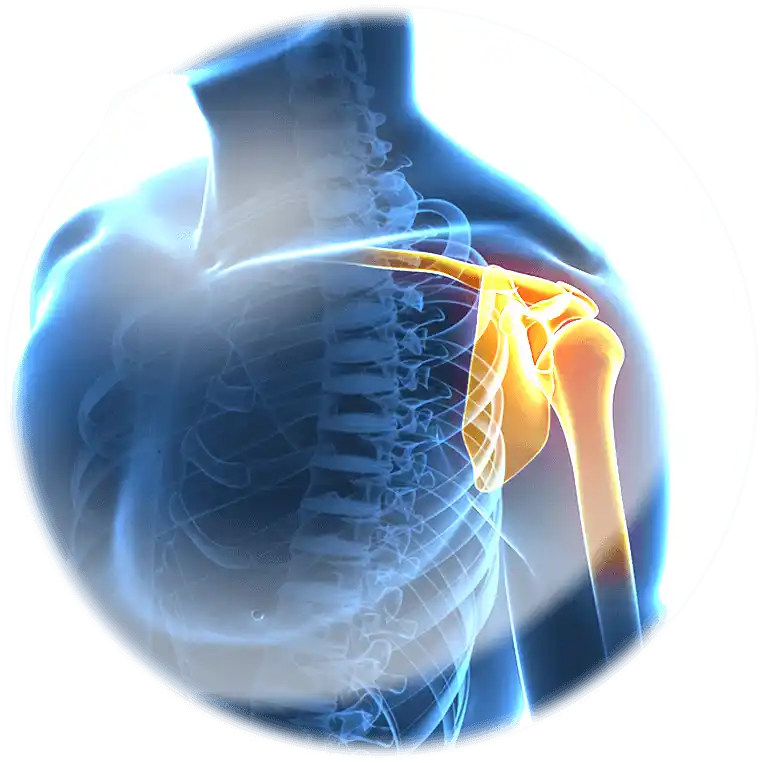

Innovative technology for release
of active ingredients
for topical and transdermal use


from Altergon Italia R&D laboratories
the new frontier of Hyaluronic Acid
"In Altergon we work steadily to increase the safety and efficacy of our products, to improve the quality of our patients life”
Our strength is in combining the strict respect of the rules and ethical principles that govern the production of the drug, with the focus on innovation and scientific research
see more on the companyUn'innovativa C.D.M.O.-Contract Development & Manufacturing Organization- per proporre moderni sistemi di Drug Delivery alle società clienti.


An important achievement that highlights the expertise and competencies of Altergon Italia: Altergon Sodium Hyaluronate has been approved by the Korean Authority (KFDA). Our identity, Your Success!
Altergon Italia is pleased to inform you that EDQM has released the renewed CEP (CEP 2014-263 – Rev 01) on 1st March 2024.
We are pleased to inform you that the EDQM has released to Altergon Italia the renewed CEP (R1-CEP- 2014-263 rev. 00) on 22th January 2021.
Altergon Italia is pleased to inform you that a revised Certificate of Suitability (RO-CEP-2014-63 Rev 01) was issued by EDQM on March 31, 2020.
In March 2016, Altergon submitted to FDA (CDRH) the Master File for manufacturing Sodium Hyaluronate (MAF)
Altergon Italia is pleased to inform that our Ultrapure Hyaluronate successfully succeeded the Certificate of Suitability (CEP: R0-CEP 2014-263-Rev 00) to the European Pharmacopoeia, Sodium Hyaluronate Monograph granted by the EDQM.
From start-up to European leader in producing transdermal medicines
pag. 73 Platinum July 2015

Please be informed that our offices will be closed for the holidays from May 30th to June 2nd, 2026. We will resume normal business operations on Tuesday, June 3rd, 2026.

PR CAMPANIA FESR 2021 – 2027
ASSE PRIORITARIO 1 BIS – TECNOLOGIE DIGITALI, PULITE E BIOTECNOLOGIE: CONTRIBUTO ALLA PIATTAFORMA STEP
ASSE 1 – Priorità di investimento 1.b – Azione 1.1.3 LDR
OBIETTIVO SPECIFICO 1.6 - SOSTENERE GLI INVESTIMENTI CHE CONTRIBUISCONO AGLI OBIETTIVI DELLA
PIATTAFORMA PER LE TECNOLOGIE STRATEGICHE PER L’EUROPA (STEP) DI CUI ALL’ART. 2 DEL REGOLAMENTO UE
2024/795
AZIONE 1.6.1 – SOSTENERE LO SVILUPPO E LA FABBRICAZIONE DI TECNOLOGIE CRITICHE
DELIBERA DI GIUNTA REGIONALE N. 481 DEL 24/09/2024
AVVISO PUBBLICO
AIUTI PER LO SVILUPPO E LA FABBRICAZIONE DI TECNOLOGIE CRITICHE – STEP
DECRETO DIRIGENZIALE N. 93 DEL 21/03/2025
La strategia alla base del progetto SAFE-GAGs si fonda su una visione integrata e a lungo termine che unisce eccellenza scientifica, innovazione industriale e risposte concrete alle tendenze e ai fabbisogni emergenti del mercato biomedicale e dermoestetico.
L’obiettivo primario del progetto è la progettazione, sviluppo e industrializzazione di una nuova generazione di filler iniettabili biotecnologici a base di glicosamminoglicani (GAGs) crosslinkati, che si distinguano per l’elevata biocompatibilità, la maggiore efficacia rigenerativa, la sicurezza d’uso e la sostenibilità del processo produttivo.
Con questo progetto la ALTERGON ITALIA SRL – UNIPERSONALE intende consolidare la leadership nel settore biomedicale, penetrare in nuovi mercati ad alto valore aggiunto, come quello della medicina rigenerativa articolare e delle applicazioni topico-mucosali, progettando una nuova generazione di filler iniettabili biotecnologici a base di glicosamminoglicani (GAGs) crosslinkati.
Link al programma: AIUTI PER LO SVILUPPO E LA FABBRICAZIONE DI TECNOLOGIE CRITICHE - STEP
Altergon Italia Srl Unipersonale ha dato il nome al Contratto di Sviluppo CDS_000463 che vede la partecipazione di importanti aziende campane attive nel settore farmaceutico. L'investimento complessivo ammissibile ammonta a 48,8 milioni di euro di cui 33,8 milioni di euro di agevolazioni concesse da Invitalia Spa, nella forma di contributo a fondo perduto e finanziamento agevolato, e prevede 22 nuovi posti di lavoro. Nello specifico Altergon Italia Srl Unipersonale sta realizzando un programma di investimento (CUP C42B15000040008) finalizzato all’ampliamento dell’attuale insediamento produttivo ubicato nella zona industriale del Comune di Morra de Sanctis (AV) con l’implementazione di nuove linee di produzione. Al contempo l’impresa è impegnata in un Progetto di Ricerca Industriale e Sviluppo Sperimentale (CUP C41B15000570008), attuato in collaborazione con l’Università degli Studi della Campania Luigi Vanvitelli - Dipartimento di Medicina Sperimentale, l’Università degli Studi di Napoli Federico II-Dipartimenti di Scienze Chimiche e di Farmacia e l’Università degli Studi di Salerno – Dipartimento di Farmacia. Gli obiettivi realizzativi in capo ad Altergon Italia Srl Unipersonale sono due: Il primo denominato “Nuove matrici per la costruzione di cerotti innovativi, garze impregnate ed altri presidi medici” ha come risultato l’individuazione dei principi attivi e delle matrici da impiegare nei cerotti a rilascio controllato, mentre il secondo denominato “Nuovi filler iniettabili” guarda al futuro con una nuova generazione di filler “intelligenti” ad alta biocompatibilità e prolungato effetto riempitivo, in grado di stimolare risposte cellulari che favoriscono la rigenerazione tessutale, contrastando in maniera attiva i segni dell’invecchiamento cutaneo.

Unione Europea
Fondo Europeo di Sviluppo Regionale
ASSE 1 – Priorità di investimento 1.b – Azione 1.1.3 LDR
Contratto di Sviluppo CDS_000463 con il Ministero dello Sviluppo
Economico Approvato con Determina Invitalia del 26.06.2019
Obblighi informativi per le erogazioni pubbliche (legge 124/2017 articolo 1 commi 125-129, modificato dal DL 34/2019): gli aiuti di Stato e gli aiuti de minimis ricevuti dalla nostra impresa sono contenuti nel Registro nazionale degli aiuti di Stato di cui all'art. 52 della L. 234/2012 e consultabili al seguente Link, inserendo come chiave di ricerca nel campo CODICE FISCALE 01367710439
Altergon Italia Srl Unipersonale è capofila di un progetto di ricerca e sviluppo dal titolo “Prodotti innovativi ad alto contenuto biotecnologico per il settore biomedicale–INBIOMED” CUP: B26G18000360005 Fonte di finanziamento: Ricerca e Innovazione 2014-2020 Riferimento decreto di concessione: D.D. del 13 luglio 2017 n. 1735 (Avviso) Importo complessivo: 6.375.080,00 euro Importo Altergon: 2.004.080,00 Tale progetto vede il coinvolgimento di altre aziende campane (o con sedi nel territorio regionale) attive nel settore della Salute, oltre a soggetti pubblici di ricerca: Tecnobios Srl - Technogenetics Spa - Università degli Studi del Sannio- Università degli Studi della Basilicata - Università degli Studi di Padova. Nello specifico Altergon Italia Srl Unipersonale realizza un programma di R&D finalizzato allo sviluppo ed implementazione di processi biotecnologici per la produzione di biopolimeri avanzati, alla realizzazione di dispositivi a base di polimeri biotecnologici per il trattamento di patologie ortopediche e per l'accrescimento di tessuti soffici e alla realizzazione dei dimostratori ed analisi delle prestazioni.
